This week, we deep dive into a paper recently published in Environmental Science: Advances. The study was led by Anass Wahby, affiliated with the Laboratory of Water, Research, and Environmental Analysis of Abdelmalek Essaadi University in Tetouan (Morocco).
This article provides a comprehensive and critical review of the latest progress in engineered biochar materials as sustainable, low-cost sorbents for carbon dioxide capture. It highlights how different synthesis and modification strategies—such as physical activation, chemical doping, and hybrid composites—shape the structural properties and CO₂ adsorption performance of biochar. A detailed comparative analysis shows that tailored biochar can rival more expensive carbon materials in CO₂ uptake, selectivity, and reusability. Notably, vanadium-oxide-modified woody biochar exhibits exceptionally high adsorption capacity and stability. The authors also identify key limitations hindering scalability and propose research directions to bridge lab insights with industrial implementation. Overall, the review positions engineered biochar as a promising platform toward sustainable CO₂ mitigation technologies.
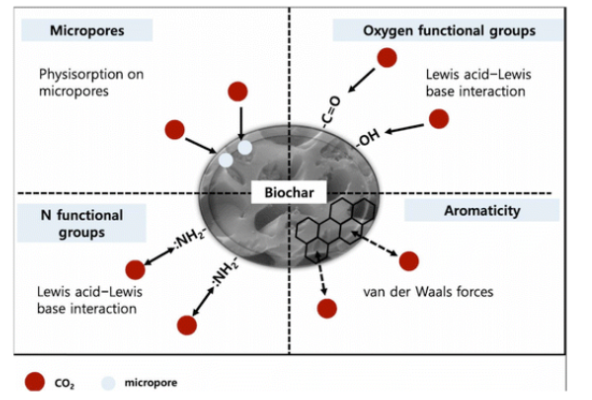
Biochar has long been recognized for its potential in environmental remediation due to its porous carbonaceous structure, eco-friendly production, and cost advantages. In this paper, the authors systematically connect synthesis pathways (e.g., pyrolysis, hydrothermal carbonization, gasification, microwave-assisted carbonization) with tunable surface properties such as porosity, surface area, chemical functionality, and basicity. This structure–property framing clarifies how specific engineering strategies—like activation with CO₂, steam, or alkali agents, metal oxide doping, and heteroatom incorporation—can dramatically enhance CO₂ adsorption mechanisms. By emphasizing measurable performance metrics (adsorption capacity under practical temperatures and pressures, CO₂/N₂ selectivity, and adsorption–desorption stability), the review highlights both well-established outcomes and emerging directions that distinguish engineered biochar from conventional pristine biochar and other synthetic carbon sorbents.
The main findings underscore several noteworthy results. Among the diverse engineered materials examined, woody biomass biochar impregnated with vanadium oxide stands out with a CO₂ adsorption capacity of ~9.8 mmol g⁻¹ and excellent recyclability over many cycles with negligible performance loss—benchmarks that position it among the top performers in physisorbent classes. A range of other modification strategies also show sizeable gains: nitrogen-doping, metal and metal oxide incorporation (e.g., Mg²⁺, CuBTC composites), and hybrid pore architectures all contribute to enhanced adsorption performance, often with improved selectivity against N₂. The review also critically discusses real-world challenges, notably moisture effects, scalability of production methods, and integration into industrial direct air capture systems, providing practical directions for bridging lab-scale insights with large-scale deployment.
Here is a list of the main takeaways of this paper:
- Engineered biochar shows strong potential as a low-cost CO₂ adsorbent with easily tunable structure and surface chemistry compared to traditional carbon materials.
Synthesis method critically determines performance: pyrolysis, activation, doping, and microwave/chemical treatments influence pore structure and adsorption capacity. - Vanadium-oxide-modified woody biochar demonstrated one of the highest CO₂ uptake values (~9.8 mmol g⁻¹) with excellent cyclic stability.
- Surface functionalization and heteroatom incorporation (e.g., N, Mg, Cu) improve CO₂ selectivity and increase basic adsorption sites.
- Challenges for industrial application include moisture sensitivity, large-scale production cost, and process integration, with strategic research needed to overcome these.
Read the full paper here: Recent advances in biochar-based engineered materials for efficient removal of CO2: from lab to industrial scale applications